Safe, quick, and painless collection
Whether you have a vaginal birth or a C-section, the process of collecting your baby’s stem cells is safe, painless, and non-disruptive to you, your baby, and your birth plan.
Give your baby a lifetime of protection by banking with Cordlife today.
Focus on the baby and we will do the rest!
After your baby is born, the umbilical cord is clamped and cut. That is when your caregiver will insert a needle into the umbilical vein to collect the remaining blood (If cord blood isn’t collected, it’s generally discarded as medical waste).
Our heparin-free collection bags are sterile and simple for your doctor to use. If you have also decided to collect the cord tissue, a segment of the umbilical cord is collected and placed into the collection jar provided. It is then sent back to our lab with the cord blood bag.
Once collection is done, your OBGYN will seal the bag, attach the pre-printed labels with your information, and place it in the Cordlife collection kit.
Please note that the final decision of whether the cord blood collection should take place will always reside with your caregiver whose first priority is the safety of you and your baby.
From your delivery room, to our lab.
After you have notified us of your delivery and reviewed the kit’s contents and pre-printed labels, a medical courier will pick up the collection kit from your hospital room and deliver it to Cordlife’s processing and storage laboratory.
We will notify you as soon as your kit arrives at our laboratory.
How is your Baby's Cord Blood Processed?
Stem cell isolation/identification is a critical step in cord blood banking. It affects the number of stem cells that can be harvested or recovered from the cord blood. Cell recovery rates are critical because a higher number of stem cells could enhance the success of the transplant or treatment. That's why Cordlife has invested today's most advanced automated stem cell processing technology which is a safe, sterile and fully automated cord blood processing technology - so you get more stem cells for maximum transplant success*.

Storing your Baby's Cord Blood
Cordlife uses U.S. FDA approved cryogenic storage pouch, with 2 integrally attached segments (20% and 80%) which are in compliance with AABB and FACT-Netcord standards. The dual integrated segments addresses the possibility for future stem cell expansion programmes. This means that when stem cell expansion technology is commercially viable, you can withdraw 80% of the stem cells for immediate use/treatment while continuing to store the 20% balance for future expansion. This storage pouch is made of a special material designed specifically to withstand cryogenic temperature at optimal cryogenic temperature of below -150°C.
The integral segments also provide the safety and assurance that additional product testing is performed on the associated unit thereby eliminating testing and cord blood unit mix ups. This is to allow for additional testing of the sample should it be required in the future, or for viability testing prior to a transplant. This storage pouch is made of a special material designed specifically to withstand cryogenic temperatures.
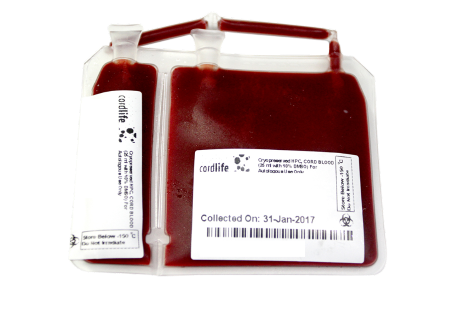
Why divide your child's precious stem cells into two cryo bags, and locations and double your risk?
Splitting the cord blood unit into multiple bags increases the risk of contamination. The unnecessary location move may heighten the risks of exposing your baby's cord blood to heat, resulting in cellular damage and possibility of sample mix-up.
The volume of a cord blood unit does not equate to cell count; splitting into multiple bags does not guarantee more stem cells. The success of a stem cell transplant largely depends on the amount of stem cells used in the treatment; splitting and storing of cord blood in multiple bags does not guarantee multiple treatments.
Given that the cell dose is an important predictor of successful transplant outcomes, such 'split CB units' might place their potential recipients at a disadvantage and their application, therefore, unlikely, unless successful stem cell expansion becomes practical in this setting.2
2Rubinstein P. Cord blood banking for clinical transplantation. Bone Marrow Transplantation. 2009; 44:635-642.
Cordlife also uses MVE Anti-contamination Vapour-phase Liquid Nitrogen Storage System for long-term stem cell cryopreservation.
Vapour-phase storage is preferred over liquid-phase storage as it eliminates the chance of cross contamination between cord blood units as compared with those stored in liquid nitrogen.
The MVE anti-contamination Vapour-phase Liquid Nitrogen Storage System operates perfectly, even without electrical supply, and is 100% reliable and fail-safe with no chance of robotic malfunction. Unlike other storage systems, our tanks have never been recalled by the U.S. FDA or the manufacturer.
Over 99% of private cord blood banks also use vapour-phase freezers.

Why do parents save their baby's umbilical cord blood, cord tissue and cord lining?

Rich source of stem cells
Your baby’s umbilical cord has the youngest and most active stem cells compared with other stem cell sources such as embryo, bone marrow, and peripheral blood.

Proven use in transplants
More than 80 diseases can potentially be treated with cord blood, and 40,000 transplants have taken place worldwide.

Untapped clinical potential
Researchers all over the world are learning how to use stem cells to treat more diseases and repair damaged tissue or organs.

Treatment options for the family
The stem cells you stored are a perfect match for your baby, and they could be used to treat other members of your family.

Readily available for a lifetime
Stem cells can be kept indefinitely if they are properly stored, and you can retrieve them whenever you need them.
